Novel process extracts rare earth elements from waste
Rare earth elements (REE), a group of 17 metallic elements, are in nearly every piece of technology, including cell phones, televisions, computers and almost every part of a vehicle. The demand for these elements increases annually, however the supply is limited geopolitically and is mined with environmentally unsustainable practices.
Young-Shin Jun, professor of energy, environmental & chemical engineering in the McKelvey School of Engineering at Washington University in St. Louis, and her team have created a proof-of-concept solution: extracting REEs from coal fly ash, a fine, powdery waste product from the combustion of coal.
“We wanted to use a greener process to extract REEs than traditionally more harmful processes,” Jun said. “Since the coal has already been used, this process is ultimately a pathway toward reduction and remediation of waste products.”
Jun and her former doctoral student, Yaguang Zhu, now a postdoctoral scholar at Princeton University, developed this novel extraction process using supercritical fluid, commonly used to decaffeinate coffee, to recover these critically needed REEs from material that would have otherwise been discarded in a landfill. Supercritical fluid is a substance at a temperature and pressure above its critical point with properties between a liquid and a gas. With more than 79 million metric tons of coal fly ash generated in the U.S. annually, Jun’s team reported that the potential value of the REEs that could be extracted from coal fly ash in the U.S. is estimated at more than $4 billion annually.
Their work, which appears in RSC Sustainability, is the first to show that common and accessible supercritical fluids, including carbon dioxide, nitrogen and air, were able to extract REEs and separate impurities very efficiently. In addition, through experiments using coal fly ash, they found that supercritical carbon dioxide decreased the concentrations of impurities in the final REE product. Ultimately, their final products contained up to 6.47% REEs, compared with 0.0234% in the initial coal fly ash source.
“The uniqueness of our work is not only using the supercritical CO2, but also showing that supercritical air and nitrogen, with much lower temperature and pressure than those required for CO2, can extract REE effectively,” said Jun, who leads the Environmental NanoChemistry Laboratory.
“We can use lower temperatures and pressures with nitrogen or air to extract the rare earth elements from coal fly ash, which means lower energy cost. Of course, the supercritical CO2 works best, but supercritical air or nitrogen could do a much better job compared with traditional high temperature boiling with acids and organic solvents for REE extraction.”
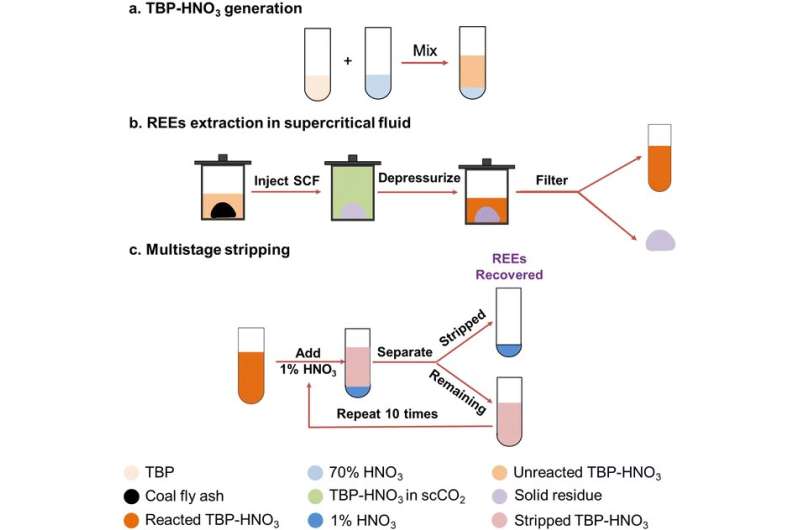
Jun’s team’s extraction process involved two steps: First, metal ions in the coal fly ash, including REEs and impurities, leach from the coal fly ash and react with nitric acid to form metal nitrates; and second, the metal nitrates react with tributyl phosphate (TBP). They found that with supercritical carbon dioxide, nitrogen or air, the REEs formed complexes that could be extracted from the coal fly ash.
After extraction, their multistage stripping process collected REEs and decreased the concentration of impurities. The nitric acid and TBP used in the process can be fully recycled multiple times without sacrificing efficiency, which minimizes their disposal concerns.
Jun’s method also eliminates the need to roast raw materials at extremely high temperatures, or greater than 500 C, and the need to extract the REEs with strong acids and a large quantity of toxic organic solvents, which also become a waste product in traditional extraction processes.
“Supercritical fluid is considered as a greener solvent, is less invasive to the environment and allows us to extract REE directly from solid waste without leaching and roasting raw materials, so less energy is required for our new process, which also produces less waste,” Jun said. “We are seeking a more environmentally benign process for critical element recycling and recovery from materials previously considered to be waste.”
More information:
Yaguang Zhu et al, Supercritical carbon dioxide/nitrogen/air extraction with multistage stripping enables selective recovery of rare earth elements from coal fly ashes, RSC Sustainability (2023). DOI: 10.1039/D2SU00033D
Washington University in St. Louis
Citation:
Novel process extracts rare earth elements from waste (2023, March 24)
retrieved 25 March 2023
from https://techxplore.com/news/2023-03-rare-earth-elements.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
Comments are closed